Researchers described a new braking mechanism in the brain that prevents normal pain from persisting for a long time, the collapse of which leads to chronic pain.
In a laboratory at the Hebrew University of Jerusalem, Alexander Binshtok, a pain neurobiologist, and his group peer into rodent brains to understand the mechanisms of pain. But that isn’t what he always did. As a young physical therapist, Binshtok worked to treat pain in patients, but he wasn’t always successful.
“I realized that although we are very good at treating acute pain, it’s almost impossible to treat chronic pain,” he said. This realization encouraged Binshtok to switch career gears and investigate what drives pain.
Now, in a new study published in Science Advances, Binshtok and his team described an inbuilt braking mechanism in the mouse brain that dampens the intensity of acute pain, preventing it from going into overdrive.1 The failure of these brakes caused chronic pain to set in. Identifying the molecular players of this regulation could open avenues for new preventive therapies for chronic pain.

Alexander Binshtok, a pain neurobiologist at The Hebrew University of Jerusalem, studies cellular and molecular mechanisms of acute and chronic pain.
Maxim Dinshtein
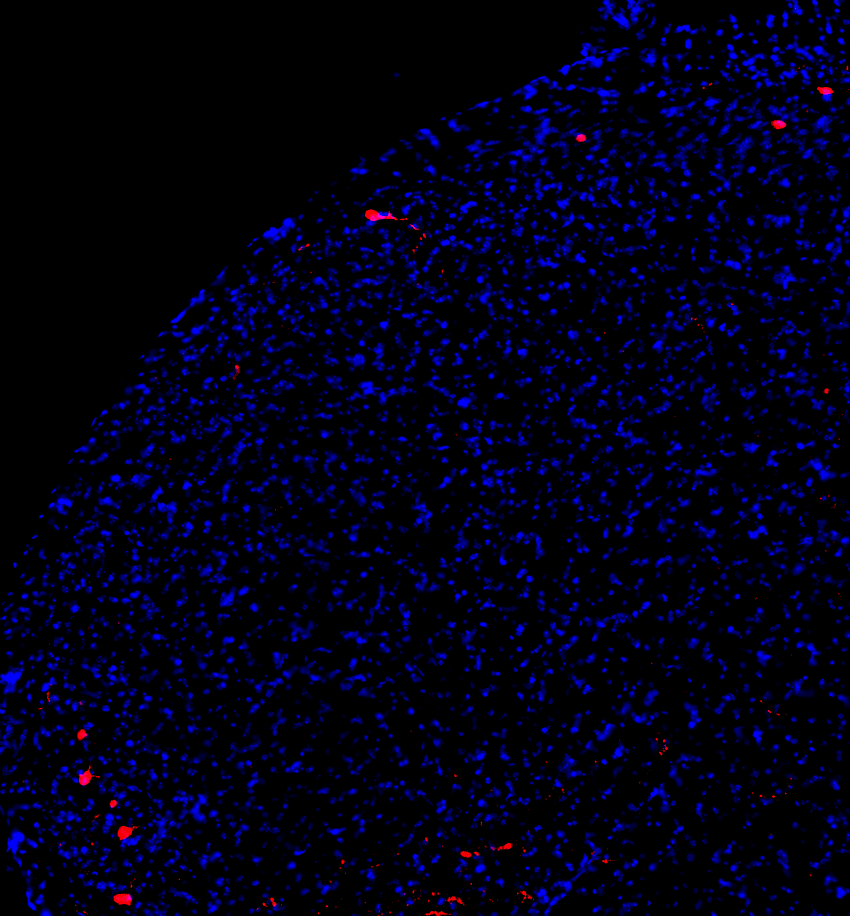
The body detects painful stimuli via primary sensory neurons, which then convey information to the pain processing region of the brainstem called the medullary dorsal horn.2 Neurons projecting from the dorsal horn to higher brain centers carry the message forward. In pathological conditions that are increasingly associated with persistent pain, such as nerve injury or inflammation, the sensory neurons become hyperexcitable, leading to enhanced activity in the dorsal horn neuronal network. Binshtok was curious to know if and how changes in the properties of the dorsal horn projection neurons (PNs) lead to chronic pain in these dysfunctional scenarios.
To test their theory, Binshtok and his team recorded the activity of dorsal horn PNs in mice that either experienced acute pain or chronic pain. The researchers used ultraviolet light to wound the animal’s cornea, thus inducing short-lived pain that lasted for about 32 hours and resolved four days after the injury. They observed higher numbers of activated dorsal horn PNs in response to this acute pain as compared to mice that were unharmed. However, contrary to the team’s expectations, the intrinsic excitability and the firing rate of the neurons dipped two days after the injury, at a time when the pain was at its peak. In essence, the neurons dampened the pain signals even though the initial stimulus was still present.
“We know that we have all kinds of endogenous controls that try and ensure that nociceptive processing is kept low, inhibited as much as possible,” said Kirsty Bannister, a pain neuroscientist at Imperial College London who was not involved in the study. She elaborated that once the brain is alerted to an injury, it tries to make sure that the body stops feeling the pain. “Here we have another wonderful example of that naturally occurring pain inhibitory control.”
But Binshtok wasn’t convinced, at least initially. “When I saw this data I said, ‘It cannot be. It’s opposite to what we all believe.’ What we know about pain is that, everything should increase,” he said. But when additional experiments produced the same results, he speculated that the reduction in neuronal excitability could be the neurons adapting to a sustained stimulus. Scientists have previously shown that neurons can tune their activity depending on the intensity and frequency of inputs they receive.3 Dialing up the input causes the neuron to tune down its output, and vice versa. “Maybe this adaptive mechanism prevents the system from going hyperactive and developing chronic pain,” he said.

Medullary dorsal horn projection neurons (red) receive information about painful stimuli from sensory neurons and transmit it to higher brain centers.
Ben Title
To establish chronic pain in the mice, the researchers mechanically constricted a branch of the trigeminal nerve, which is a nerve in the face, and induced pain in it that lasted for weeks. Upon recording the activity of dorsal horn PNs two to four weeks after the procedure, they observed increased neuronal activity. There was no change in the internal excitability of the neurons in the early or late stages of the chronic model, which prevented the neurons from modulating their output. Unlike the acute pain conditions, the projection neurons in the chronic model were unable to employ brakes to control their output, causing relentless pain.
“It’s incredibly interesting that these very neurons that convey nociceptive information change their intrinsic properties according to the injury,” Bannister said.
Further, Binshtok and his colleagues observed that an increase in the projection neurons’ potassium A-current, which can modulate a neuron’s activity patterns, contributed to the change in their excitability under acute pain conditions. No such current alterations occurred in the chronic model.
Binshtok now wants to understand the cellular, molecular, and biophysical mechanisms that drive the brakes in acute pain and why they fail in chronic pain. “If we can find the difference and funnel [it] to a specific molecule, we can then use it as a target to prevent the transition from acute to chronic pain,” he said.
Source link




